Translate this page into:
Development of a controlled release formulation of an indigenous insect growth regulator, DPE-28, a substituted diphenylether, for controlling the breeding of Culex quinquefasciatus
Reprint requests: Dr M. Kalyanasundaram, Scientist ‘G’, Vector Control Research Centre (ICMR), Medical Complex, Indira Nagar, Puducherry 605 006, India e-mail: ananthikannan@ymail.com
-
Received: ,
This is an open-access article distributed under the terms of the Creative Commons Attribution-Noncommercial-Share Alike 3.0 Unported, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This article was originally published by Medknow Publications and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Background & objectives:
DPE-28, a substituted diphenyl ether (2,6-ditertiarybutyl phenyl-2’,4’-dinitro phenyl ether) was reported to exhibit promising insect growth regulating activity against Culex quinquefasciatus, the vector of lymphatic filariasis. A controlled release formulation (CRF) of DPE-28 has been developed to control Cx. quinquefasciatus in its breeding habitats. Toxicity of DPE-28, safety to non-target mosquito predators and the release profile of the CRF of DPE-28 are studied and discussed.
Methods:
The acute oral and dermal toxicity was tested in male and female Wistar rats as per the Organization for Economic Cooperation and Development (OECD) guidelines 425 and 402 respectively. The toxicity of DPE-28 to non-target predators was tested as per the reported procedure from this laboratory. The CRF of DPE-28 was prepared by following the reported procedure developed at this laboratory earlier. The concentration of DPE-28 released from the CRF was monitored by HPLC by constructing a calibration graph by plotting the peak area in the Y-axis and the concentration of DPE-28 in the X-axis.
Results:
DPE-28 has been tested for acute oral toxicity and found to be moderately toxic with LD50 value of 1098 mg/kg body weight (b.w). The results of the acute dermal toxicity and skin irritation studies reveal that DPE-28 is safe and non-irritant. DPE-28 when tested at 0.4 mg/litre against non-target mosquito predators did not produce any mortality. The release profile of the active ingredient DPE-28 from the CRF by HPLC technique showed that the average daily release (ADR) of DPE-28 ranged from 0.07 to 5.0 mg/litre during first four weeks. Thereafter the matrix started eroding and the ADR ranged from 5 to 11 mg/litre during the remaining 5 wk. The cumulative release of active ingredient showed that > 90 per cent of the active ingredient was released from the matrix.
Interpretation & conclusions:
The controlled release matrix of DPE-28 was thus found to inhibit the adult emergence (>80%) of Cx. quinquefasciatus for a period of nine weeks. The CRF of DPE-28 may play a useful role in field and may be recommended for mosquito control programme after evaluating the same under field conditions.
Keywords
Controlled release formulation
diphenylether
DPE-28
insect growth regulator
mosquito larval control
The environmental hazards posed by conventional mosquito larvicides coupled with the development of resistance in mosquito vectors to some of these insecticides warrant an alternate approach in the chemical control of vectors. The insect growth regulators (IGRs) represent a major breakthrough in insecticide chemistry and much work has been done during the past three decades. IGRs are generally classified into two groups1 according to the mode of action. Some compounds are known to inhibit the formation of chitin, which is required for moulting from one stage into the next stage and these compounds are known as chitin synthesis inhibitors and juvenoids are the analogues of juvenile hormones that inhibit pupation.
A large number of synthetic chemicals possessing insect growth regulating property have been found to be promising in controlling insects of public health importance, such as mosquitoes, chironomid midges and pestiferous flies2–4. Compounds belonging to various chemical classes such as amides5, oxime ethers6 and carbamates7 have been earlier shown to possess promising insect growth regulating activity against various mosquito vectors. IGR activity against scale insects was studied with a phenoxyphenoxy carbamate8. A pyridyl terpenoid ether has also been studied for IGR activity against mosquitoes9. Pyriproxifen [4-(phenoxyphenyl)-(RS)-2-(2-pyridyloxy) ethyl ether] has been evaluated as a 0.5 granular (GR) formulation in field for controlling the breeding of Cx. quinquefasciatus10. Recently, a benzoylphenylurea derivative, novaluron11, was reported to have promising IGR activity for controlling Cx. quinquefasciatus in various breeding sites.
Earlier, the structure-activity relationships in diphenylethers for insect growth regulating activity against mosquitoes were studied and one of the compounds, DPE-28 (2,6-ditertiarybutyl phenyl-2’,4’-dinitro phenyl ether) was found to be highly effective against Cx. quinquefasciatus with EC50and EC90(concentration to inhibit 50 and 90% of adult emergence) values of 2.2 × 10-3and 1.16 × 10-2 mg/litre respectively12. It was earlier reported13 that the treatment of mosquito eggs with an EC50 dose of DPE-28 caused mortality while an EC90 dose completely ceased adult emergence. There was a significant reduction in the fecundity and fertility rates of emerged mosquitoes when treated with EC50 dose of DPE-28. Studies on the effect of the juvenoid, DPE-28 on the biology and behaviour of Cx. quinquefasciatus showed that the developmental duration, sex ratio, mating success and blood feeding were considerably affected by the exposure of larvae and pupae to this compound14. The present investigation deals with studies on the toxicological data, safety to various non-target organism and development of an environment-friendly controlled release formulation15 and the release kinetics of the active ingredient from the matrix under simulated conditions.
Material & Methods
Synthesis of the diphenylether DPE-28: The diphenylether, DPE-28 was synthesized by a procedure reported from this Centre earlier12.
Toxicological studies on DPE-28: Toxicological studies on DPE-28 were carried out at International Institute of Biotechnology and Toxicology (IIBAT), Padappai, Tamil Nadu after obtaining the approval of the Ethical Committee of IIBAT. The acute oral toxicity of DPE-28 was tested in female Wistar rats by following the Organization for Economic Cooperation and Development (OECD) guideline 42516. In main test dose progression 1.75, 5.5, 17.5, 55, 175, 550 and 2000 mg/kg b.w. doses were selected as per the guideline. Following dosing, the rats were observed for 14 days for mortality and clinical signs of toxicity. Bodyweight gains of the survived animals were noted weekly. A gross necropsy was performed on all animals that died during the study and on all survivors on day 14.
The acute dermal toxicity of DPE-28 was tested in male and female Wistar rats as per the OECD guideline 40217. Appropriate weight of the test substance was evenly applied at the dose of 2000 mg/kg b.w. to the clipped area of skin of the animals. Following dosing, the rats were observed for 14 days for mortality and clinical signs of toxicity. Bodyweight gain was noted. All animals were necropsied at the end of the 14-day observation period.
The primary skin irritation potential of DPE-28 was tested in female New Zealand white rabbits as per OECD guideline 404. A quantity of 0.5 g of the test substance was moistened with minimum volume of distilled water so as to prepare a paste. The paste thus prepared was evenly applied to the clipped area of the skin of the rabbits under a gauze patch. At the end of 4 h, the gauze patch was removed and application site was wiped with water without altering the integrity of the epidermis to remove the residual test substance. The experiment was repeated to confirm the non-irritant response of the test substance by using two additional animals. The animals were observed for erythema and oedema at 60 min, 24, 48 and 72 h following the removal of the gauze patch.
Effect on non-target organism: The effect of DPE-28 at 0.4 mg/litre (> 30 times the EC90 value of the compound to Cx. quinquefasciatus) was studied on non-target mosquito predators like, fish (Gambusia affnis), water bug (Diplonychus indicus) adults, dragon fly (Anisoptera species) nymphs, water scorpion (Nepa species) adults and tadpole (Rana hexadactyla) by following the reported procedure from this laboratory18. One predator was released into 250 ml disposable cup containing 100 ml tap water. Only one predator was used so as to avoid cannibalism. Ten replicates were performed for the test concentration along with four replicates of untreated controls. The predators were observed for mortality after every 24 h exposure period for 72 h.
Controlled release formulation of DPE-28: A 20 per cent emulsifiable concentrate (EC) was made by using xylene as solvent and tween 80 as emulsifier and used for development of a controlled release formulation. An aqueous (20%) slurry of sodium carboxymethylcellulose (NaCMC) (Commercial grade, as a 10% aqueous slurry gives a viscosity of 65 dPa at 28°C) was made homogeneous by stirring with 1 per cent expanded polystyrene beads as floating agent and required quantity of 20 per cent EC. The quantity of EC was adjusted in such a way that each pellet would contain 1 g of the active ingredient (ai). The slurry was transferred to special moulds for making the pellets crosslinking with an aqueous solution of aluminium sulphate (LR grade, 0.05M) with pH adjusted of 3.2 using 50 per cent NaOH for 48 h (Effective crosslinking period). The gelling medium may be reused for next crosslinking. The pellets were washed with water to remove the remaining gelling medium and dried either at room temperature for one to two weeks or at 50-55°C in a vacuum oven for 1 - 2 days. Controlled release matrices without DPE-28 were also made to serve as control matrices.
HPLC analysis for determining the concentration of DPE-28: HPLC system (ThermoFinnigan, CA) composed of Spectra System P4000 solvent delivery system, Spectra System AS3000 autosampler and Photodiode array (PDA) detector SN4000 was utilized. The output signals were monitored with ChromQuest 4.0 Chromatography workstation. HPLC analysis for DPE-28 was carried out under the following chromatographic conditions – acetonitrole / water (70:30) with 0.01 per cent phosphoric acid adjusted to pH 3.2 and at a flow rate of 1.0 ml/min. A Supelcosil C8 column (15 cm × 4.6 mm) of 3 μm particle size was used for the analysis. Analysis was done at UV 270 nm and 40°C.
Release of DPE-28 from the controlled release matrices: One controlled release matrix containing DPE-28 was released in 1.0 litre of water taken in a 2-litre beaker. Five replicates were set up for matrices with DPE-28 and five replicates were set up with matrices without DPE-28 to serve as control. Water was changed at daily intervals. Everyday 500 ml of water was withdrawn for conducting bioassay (two replicates with 250 ml for each replicate) against early third instar larvae of Cx. quinquefasciatus obtained from the cyclic colony maintained at the Centre. About 15 ml of water sample was withdrawn from each replicate, extracted with 25 ml of dichloromethane in a 50 ml separating funnel and the bottom layer was withdrawn and dried over anhydrous magnesium sulphate. This was filtered through a Whatman filter paper No.1 and taken in a 50 ml test tube and the solvent was removed in a water bath. The residue was taken up in 1 ml of acetonitrile (HPLC grade) and taken in the HPLC sample injection vial.
Results & Discussion
Acute oral toxicity: None of the animals died in groups treated with DPE-28 with 175 and 55 mg/kg b.w. for acute oral toxicity during the study period, whereas four animals died in groups treated with 2000 mg/kg b.w. The survived animals showed normal body weight gain on days 7 and 14 compared with the zero day body weight. Gross pathology examination conducted on survived animals at the end of the experimental period and animals that died during the study did not reveal any macroscopic lesion that could be attributed to the toxicity of the test material, DPE-28. Based on the experimental results, estimated acute oral LD50 of DPE-28 for Wistar rat (female) was 1098 mg/kg b.w.
Acute dermal toxicity: No clinical signs of toxicity or mortality were observed in animals treated with DPE-28 at the dose of 2000 mg/kg b. w. throughout the observation period. All the animals showed a normal trend in the body weight gain on days 7 and 14. No compound related lesions were observed under gross pathological examination. From the experimental results, acute dermal LD50 of DPE-28 for Wistar rat was considered as >2000 mg/kg b.w.
Primary skin irritation test: None of the animals exhibited any skin reaction at any of the time points of observation. Thus, the primary skin irritation index was calculated as zero. Hence it is concluded that DPE-28 was non-irritant to the skin of New Zealand white rabbits under the experimental conditions tested.
The results of the acute oral toxicity of DPE-28 to Wistar rats suggest that this compound can be considered as a moderate toxic material based on the classification of World Health Organization19. The results of the dermal toxicity and skin irritation studies reveal that this compound is safe and non-irritant.
Effect on non-target organisms: DPE-28 when tested at 0.4 mg/litre against mosquito predators, fish (Gambusia affinis), water bug Diplonchus indicus (adults), dragon fly (Anisoptera species) nymphs, water scorpion (Nepa species) adults and tadpole (Rana hexadactyla) did not produce any mortality. DPE-28 was found to be non toxic to all the five predators at a concentration of 0.4 mg/litre which is more than 30 times its EI90 value to the target mosquito vector, Cx. quinquefasciatus.
Standardization of DPE-28 and release profile of DPE from the CRF by HPLC: HPLC analysis for DPE-28 revealed that there was a peak for DPE-28 with a retention time of 5.84 min (Fig. 1). A calibration curve (Fig. 2) was constructed by plotting the peak area in the Y-axis and the concentration of DPE-28 in the X-axis. The unknown concentrations were obtained by solving the linear regression equation for the calibration curve y = 22044x + 34582.
- HPLC chromatogram of DPE-28.
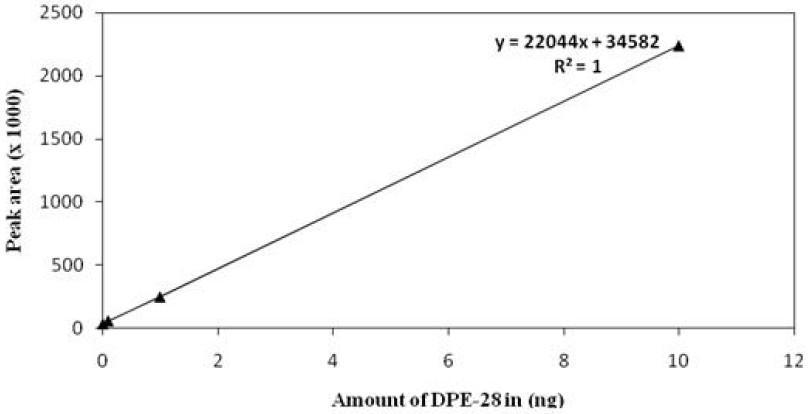
- Calibration graph for the estimation of DPE-28.
The release profile of the active ingredient DPE-28 from the controlled release matrix giving the average daily release (ADR) in mg/litre, cumulative release of the active ingredient at weekly intervals is given in Fig. 3. From the results, it was inferred that the average daily release of DPE-28 ranged from 0.07 to 5.0 mg/litre during first four weeks. Thereafter the matrix started eroding and the ADR ranged from 5 to 11 mg/litre during the remaining 5 wk. The matrix thus could inhibit the adult emergence (>80%) of Cx. quinquefasciatus during the period of nine weeks. The cumulative release of the active ingredient showed that > 90 per cent of the active ingredient was released from the matrix. This observation of a CRF exhibiting inhibition in adult emergence for a period of more than two months could be compared to the efficacy of methoprene, a widely used juvenile hormone mimic, formulated as 30-day slow release Altosid pellets for controlling mosquitoes in underground storm water drainage catch basins. The results showed that methoprene applied at 3.5 g of the pellet per basin could effectively control mosquitoes for a month under field conditions20. Similar observation was also made during the development of controlled release matrices for triflumuron, an IGR which were stable for 14 to 16 wk21. Thus, the use of controlled release formulations will be more practical and desirable and may make the Programme more cost-effective.
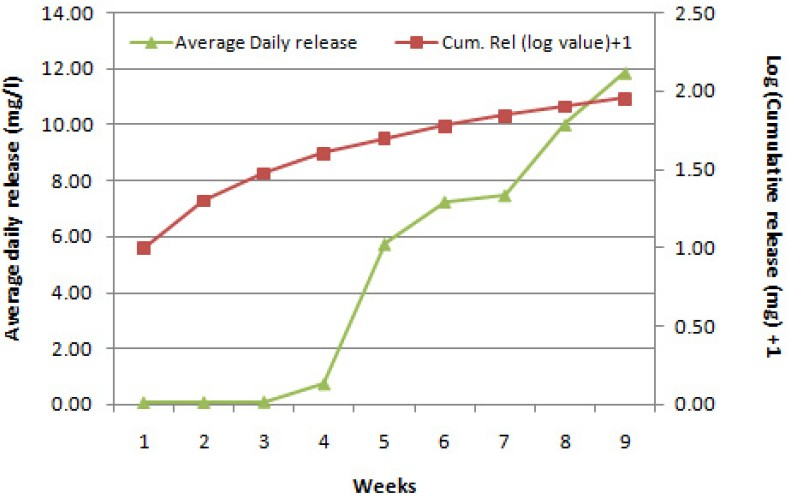
- Release profile of DPE-28 from the controlled release matrix.
The treated larvae and pupae showed various abnormalities. Larvae were usually dechitinized, elongated and attached to the previous moult. Mortality in the larval-pupal intermediate was prominent with DPE-28. The pupae formed after treatment became pale resulting in mortality. Considering the effect of the CRF of DPE-28 in inhibiting the adult emergence of Cx. quinquefasciatus for about nine weeks, its safety to non target aquatic predatory organisms tested and the other toxicological data, the CRF of DPE-28 may play useful role in field and may be recommended for mosquito control programme after evaluating the formulation in controlling Cx. quinquefasciatus in its breeding habitats.
The authors thank the Director, IIBAT, Padappai, Tamil Nadu, India for generating toxicological data on DPE-28. The technical assistance provided by Shrimati R. Anilakumari, Laboratory Technician and Shri S. Srinivasan, Laboratory Assistant of the Unit of Chemistry is also gratefully acknowledged.
Conflict of Interest: Authors declare that there is no conflict of interest.
References
- Lutte contre les vecteurs et nuisances en santé publique. Ency Med Chir Paris Maladies Infectieuses. 1980;8120(B10):3-1980.
- [Google Scholar]
- Field tests of insecticides and insect growth regulators for the control of Culex quinquefasciatus in anaerobic animal waste lagoons. Mosq News. 1980;40:36-42.
- [Google Scholar]
- Evaluation of the IGR larvadex as a feed through treatment for the control of pestiferous flies on poultry ranches. J Econ Entomol. 1983;76:515-9.
- [Google Scholar]
- Insect growth regulators for the control of aquatic midges. J Econ Entomol. 1974;67:165-70.
- [Google Scholar]
- Evaluation of a new compound (VCRC/INS/A-23) with juvenile hormone activity against mosquito vectors. Indian J Med Res. 1985;82:9-13.
- [Google Scholar]
- Oxime ethers – a new potent insect growth regulators. Agric Biol Chem. 1985;49:3197-202.
- [Google Scholar]
- Evaluation of three formulations of a chitin synthesis inhibitor (fenoxycarb) against mosquito vectors. Indian J Med Res. 1987;85:161-7.
- [Google Scholar]
- Synthesis of pyridyl terpenoid ether analogs of juvenile hormone. Agric Biol Chem. 1980;44:2709-15.
- [Google Scholar]
- Effect of a new insect growth regulator R013-5223 on scale insects. Phytoparasitology. 1982;10:27-31.
- [Google Scholar]
- Field evaluation of an insect growth regulator, pyriproxyfen 0.5% GR against Culex quinquefasciatus, the vector of Bancroftian filariasis in Pondicherry, India. Acta Trop. 2008;107:20-4.
- [Google Scholar]
- Efficacy of novaluron against Culex quinquefasciatus in small and medium scale trials, India. J Am Mosq Control Assoc. 2009;25:315-22.
- [Google Scholar]
- Structure-activity relationships in diphenyl ethers for insect growth regulating activity against mosquitoes. Indian J Med Res. 1989;89:344-9.
- [Google Scholar]
- Comparative efficacy of aphid extracts and some juvenoids against the development of mosquitoes. J Am Mosq Control Assoc. 1992;8:247-51.
- [Google Scholar]
- Effects of a juvenoid, DPE-28, on the biology and behavior of Culex quinquefasciatus, the human filariasis vector. Indian J Exp Biol. 2000;38:687-91.
- [Google Scholar]
- A process for the preparation of a controlled release agent for controlling the growth of insects. Indian Patent 191820 2005
- [Google Scholar]
- OECD guidelines for testing of chemicals (Section 4, No. 425, adopted 23rd March 2006) Paris: Organisation for Economic Co-operation and Development; 2006.
- [Google Scholar]
- OECD guidelines for testing of chemicals (Section 4, No. 402, adopted 24th February 1987) Paris: Organisation for Economic Co-operation and Development; 1987.
- [Google Scholar]
- Laboratory evaluation of methanolic extract of Atlantia monophylla (Family: Rutaceae) against immature stages of mosquitoes and non-target organisms. Mem Inst Oswaldo Cruz. 2004;99:115-8.
- [Google Scholar]
- World Health Organization. The WHO recommended classification of pesticides by hazard and guidelines to classification 2000-01 2000 WHO/PCS/01.5
- [Google Scholar]
- Efficacy of methoprene for mosquito control in storm water catch basins. J Am Mosq Control Assoc. 2006;22:333-8.
- [Google Scholar]
- Development of biodegradable aluminum carboxymethylcellulose matrices for mosquito larvicides. Pest Manag Sci. 2004;60:685-90.
- [Google Scholar]






