Translate this page into:
Emergence of integron borne PER-1 mediated extended spectrum cephalosporin resistance among nosocomial isolates of Gram-negative bacilli
Reprint requests: Dr Amitabha Bhattacharjee, Department of Microbiology, Assam University, Silchar 788 011, Assam, India e-mail: ab0404@gmail.com
-
Received: ,
This is an open-access article distributed under the terms of the Creative Commons Attribution-Noncommercial-Share Alike 3.0 Unported, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Background & objectives:
Pseudomonas extended resistant (PER) enzymes are rare type of extended-spectrum beta lactamases (ESBLs) that confer third generation cephalosporin resistance. These are often integron borne and laterally transmitted. The aim of the present study was to investigate the emergence of integron borne cephalosporin resistant PER-1 gene in diverse incompatibility (Inc) group plasmids among Gram-negative bacteria.
Methods:
A total of 613 consecutive, non-duplicate, Gram-negative bacteria of Enterobacteriaceae family and non-fermenting Gram-negative bacteria were isolated from different clinical specimens during a period of 18 months. For amplification and detection of blaPER, multiplex PCR was done. For understanding the genetic environment of blaPER-1, integrase gene PCR and cassette PCR (59 be) was performed. Gene transferability experiment was carried out and PCR based replicon typing was performed for incompatibility group typing of plasmids using 18 pairs of primers. An inhibitor based method was used for phenotypic detection of intrinsic resistance.
Results:
Multiplex PCR and sequencing confirmed that 45 isolates were harbouring blaPER-1. Both class 1 and class 2 integrons were observed among them. Integrase and cassette PCR (59 be) PCR results confirmed that the resistant determinant was located within class 1 integron. Transformation and conjugation experiments revealed that PER-1 was laterally transferable and disseminated through diverse Inc plasmid type. Efflux pump mediated carbapenem resistance was observed in all isolates. All isolates belonged to heterogenous groups.
Interpretation & conclusions:
This study demonstrates the dissemination of cephalosporins resistant, integron borne blaPER-1 in hospital setting in this part of the country and emphasizes on the rational use of third generation cephalosporins to slow down the expansion of this rare type of ESBL gene.
Keywords
ESBLs
gene cassette
Gram-negative
Inc group
integron
PER
plasmids
Pseudomonas extended resistant (PER) beta-lactamase belongs to class A extended spectrum beta-lactamase (ESBL), which was first detected in France in Pseudomonas aeruginosa isolated from a Turkish patient in 19931. The blaPER-1 gene has been detected in P. aeruginosa2, Acinetobacter spp.3, Proteus mirabilis4, and in Salmonella enterica serovar Typhimurium5. It has been reported that the genetic location of blaPER-1 is either on the chromosome or on the plasmid16. Integrons also act as small mobile elements which carry and transfer antibiotics resistant genes. The prevalence of integron borne PER-1 is very high because these carry different antibiotics resistant genes in their gene cassettes78910. This determinant is often carried by transposable elements which are responsible for their lateral spread among diverse group of bacterial pathogens2.
This study was carried out to determine the occurrence of cephalosporin resistant, integron borne blaPER-1 and its transmission dynamics in Gram-negative bacteria.
Material & Methods
Bacterial isolates: A total of 613 consecutive, non-duplicate, Gram-negative bacteria consisting of members of Enterobacteriaceae family and non-fermenting GramGram-negative bacteria were isolated from different clinical specimens during a period of 18 months (November 2012 to April 2013) from different wards/clinics of Silchar Medical College and Hospital, Assam, India. The work was performed in the department of Microbiology, Assam University, Silchar, Assam. All the organisms were subcultured on MacConkey agar (Hi-Media, Mumbai, India).
Phenotypic detection of ESBL: All isolates were subjected to preliminary screening on Mueller- Hinton (M-H) agar plates containing two antibiotics namely cefotaxime and ceftazidime at 1 µg/ml11. All positive isolates were confirmed by combined disc diffusion method using cefotaxime (30 µg) and ceftazidime (30 µg) alone and in combination with clavulanic acid (10 µg) (Hi-Media, Mumbai, India) as per Clinical and Laboratory Standards Institute (CLSI) recommendation11.
Molecular characterization blaESBL genes by multiplex PCR: For amplification and characterization of blaESBL genes, a set of five primers were used namely: blaTEM, blaCTX-M, blaSHV, blaOXA-2, and blaPER12. Reactions were run under the following conditions: initial denaturation at 94°C for five min, 33 cycles of 94 °C for 35 sec, 51°C for one min, 72°C for one min and final extension at 72°C for seven min.
Sequencing of blaESBL: PCR product was purified by Gene Jet PCR purification kit (Thermoscientific, Lithuania). In brief, 30 µl of purified products were used for sequencing along with 20 µl of blaESBL gene primers12 (10 pmol each primers). Sequence results were analyzed using BLAST suite program of NCBI (http://blast.ncbi.nlm.nih.gov/Blast.cgi).
Characterization of integrons by integrase gene PCR: For characterization of class 1 and class 2 integrons, integrase gene PCR assays were performed by using primers Int1F (CAG TGG ACA TAA GCC TGT TC), Int1R (CCC GAG GCA TAG ACT GTA) and Int2F (TTG CGA GTA TCC ATA ACC TG), Int2R (TTA CCT GCA CTG GAT TAA GC)13. Reactions were run under the following conditions: initial denaturation at 94°C for five min, 32 cycles of 94 °C for 35 sec, 51°C for one min, 72°C for one min and final extension at 72°C for seven min.
Association of blaESBL with gene cassette: For study of the association of gene cassette with blaESBL gene, 59 be cassette PCR was performed. Two PCR reactions were performed, one with HS287 and blaPER reverse, another with HS286 and blaPER forward1214. The amplified PCR products were further sequenced. PCR amplification was performed using 30 µl of total reaction volume. PCR reactions were run under the following conditions: initial denaturation at 95°C for two min, 30 cycles of 95 °C for 20 sec, 52°C for 45 sec, 72°C for one min and final extension at 72°C for five min.
Plasmid preparation: All ESBL positive bacterial isolates were cultured in Luria-Bertani broth (Hi-Media, Mumbai, India) containing 1µg/ml of cefotaxime. Cultures were incubated on shaker incubator overnight at 37°C. Plasmids were purified by Gene Jet plasmid Miniprep kit (Thermoscientific, Lithuania).
Gene transferability of blaESBL gene by transformation and conjugation: Transformation was carried out using Escherichia coli JM107 (Fermentas, USA) as recipient15. Transformants were selected on cefotaxime (0.5 µg/ml) containing L-B agar plates. L-B agar control plate was used, with and without cefotaxime 0.5 µg/ml.
Conjugation experiments were carried out between clinical isolates as donors and an streptomycin resistant E. coli recipient strain B (Bangalore Genei, Bengaluru), Overnight culture of the bacteria was diluted in Luria-Bertani broth and was grown at 37 °C till the OD of the recipient and donor culture reached 0.8-0.9 at A600. Donor and recipient cells were mixed at 1:5 donor-to-recipient ratios and transconjugants were selected on cefotaxime (0.5 µg/ml) and streptomycin (800 µg/ml) agar plates.
Plasmid profiling and incompatibility (Inc) typing of ESBL producers: For plasmid profiling, 1.5 µl of each transformants was used and analyzed by agarose gel electrophoresis (1% agarose, Hi-Media, Mumbai, India).
For detection of incompatibility group plasmid in all blaPER producing strains, PCR based replicon typing was carried out targeting 18 different replicon types, to perform five multiplex and three simplex PCRs to amplify the FIA, FIB, FIC, HI1, HI2, I1-Ig, L/M, N, P, W, T, A/C, K, B/O, X, Y, F and FIIA replicons16.
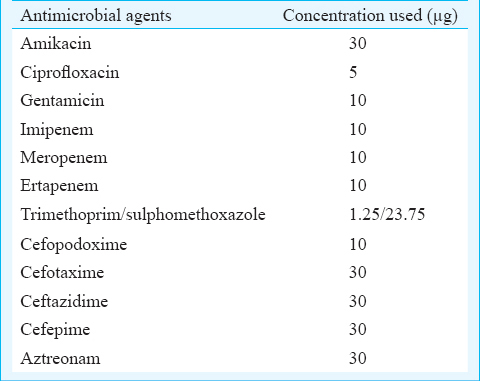
Antimicrobial susceptibility and minimum inhibitory concentrations (MIC): Antimicrobial susceptibility was determined by Kirby Bauer disk diffusion method on M-H agar plates17. Antibiotics used in this study were listed in Table I. MICs of various antibiotics were also determined on Muller- Hinton agar plates containing 2, 4, 8, 16, 32, 64, 128, 256 µg/ml of antibiotics, by agar dilution method according to CLSI guidelines11.
Detection of efflux pump activity: Efflux pump activity of the isolates were phenotypically detected by double disc synergy test using meropenem (10 µg) alone and in combination with CCCP (100 mM) (carbonyl cyanide m-chlorophenylhydrazone) (Hi-Media, Mumbai, India) as described earlier18. MIC reduction assay was performed with imipenem, meropenem and ertapenem alone and in combination with CCCP at a fixed concentration of 20 μg/ml19.
DNA fingerprinting by enterobacterial repetitive intergenic consensus (ERIC) sequences PCR: Typing of all ESBL producing isolates were done by ERIC-PCR, using two primers: ERIC-F and ERIC-R20.
Results & Discussion
Forty five isolates showed amplification with blaPER primers (Figure, Table II) which were further confirmed by sequencing as blaPER-1. However, 12 isolates were found to carry only blaPER, while in the remaining 33 co-existence of CTX-M-15, SHV-148 and TEM-1 was observed along with blaPER-1 (Table II).
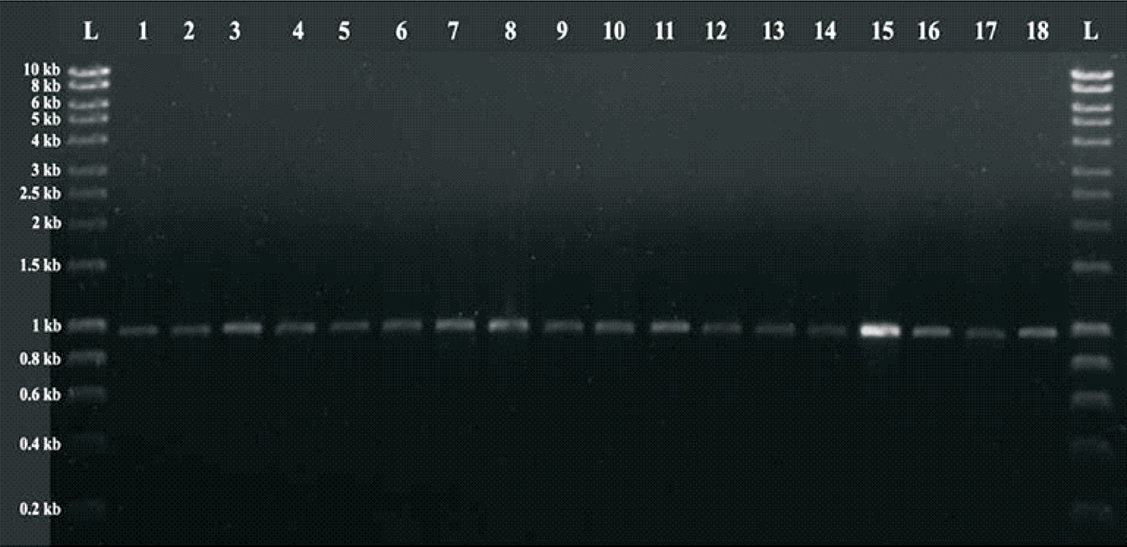
- PCR amplified product of blaPER-1. L: Hyper ladder I (Bioline; UK); 1-18: 920 bp PCR amplified product of blaPER-1.
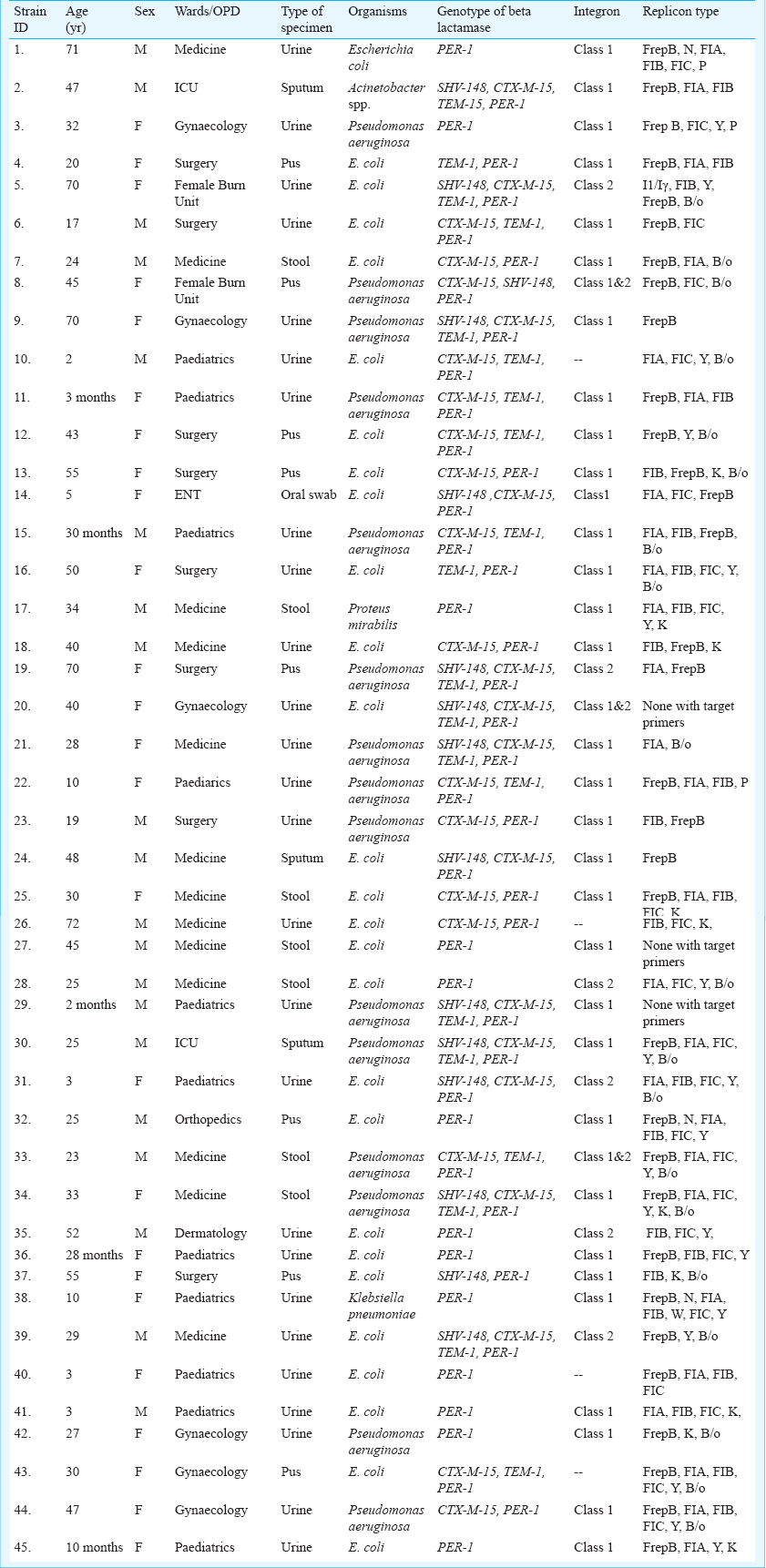
Integrase gene PCR results demonstrated that 32 isolates harboured class I, and six isolates carried class II integron. Both class I and class II integrons were found in three isolates (Table II), four isolates were negative for integron. blaPER-1 was found to be class 1 integron borne that was further confirmed by sequencing.
Transformation experiment established the fact that blaPER-1 was carried within plasmid and was conjugatively transferable in E. coli JM 107. PER-1 could be amplified by PCR in both transformants and tranconjugants. Plasmid incompatibility typing of all blaPER-1 producers illustrated that there were diverse Inc groups: N, FIA, FIB, W, Y, P, FIC, F and K were present in all blaPER-1 positive isolates (Table II).
All isolates were susceptible to tigecycline (100%) followed by imipenem (95.55%), and less susceptible to meropenem (75%). Other antibiotics like amikacin, gentamicin, ciprofloxacin, co-trimoxazole, cefepime, ceftriaxone and cefoxitin showed moderate to lower activity. A high MIC was noticed against all tested cephalosporins (≥256 µg/ml), and monobactam (≥256 µg/ml). Efflux pump mediated carbapenem resistance was noticed in all 45 isolates. A reduction in MIC was observed in all 45 isolates against imipenem, ertapenem and meropenem when CCCP was added at a fixed concentration of 20 µg/ml. All isolates were clonally non related as observed in ERIC-PCR.
In the current study blaPER-1 was found to be disseminated among members of Enterobacteriacae and in non-fermenting Gram-negative bacteria that were resistant to multiple groups of antibiotics. In Iran, the prevalence rate of PER-1 was 27.5 per cent in P. aeruginosa whereas in A. baumannii prevalence rate was 51 per cent due to indiscriminate consumption of antibiotics by patients78.
The present study gives an insight in to horizontal transferability of this resistance determinant through integron gene cassettes in Gram-negative bacteria which has also been reported from other parts of the world7910. The most important finding of this study was carriage of blaPER-1 within different incompatibility (Inc) groups of plasmids which revealed that multiple carrier vehicles were responsible for expansion of this determinant and probably had diverse source of acquisition. To complicate the situation further, carbapenem therapy is restricted due to efflux pump activity, leaving little therapeutic alternatives. The study highlights the presence of this rare type of ESBL in this part of the country and deserves immediate awareness and controlled use of broad spectrum antibiotics to slow down the maintenance and persistence of this resistant determinant within hospital environment.
Acknowledgment
The authors acknowledge the University Grants Commission and Department of Biotechnology, New Delhi for financial assistance; and Assam University Biotech Hub for providing laboratory facility.
References
- Characterization of a novel extended-spectrum β-lactamase from Pseudomonas aeruginosa. Antimicrob Agents Chemother. 1993;37:962-9.
- [Google Scholar]
- Detection of Pseudomonas aeruginosa isolates of the international clonal complex 11 carrying the blaPER-1 extended-spectrum β-lactamase gene in Greece. J Antimicrob Chemother. 2012;67:357-61.
- [Google Scholar]
- Emergence of PER and VEB extended-spectrum β-lactamases in Acinetobacter baumannii in Belgium. J Antimicrob Chemother. 2006;58:178-82.
- [Google Scholar]
- Multifocal detection of multidrug-resistant Pseudomonas aeruginosa 0producing the PER-1 extended spectrum â-lactamase in northern Italy. J Clin Microbiol. 2004;42:2523-9.
- [Google Scholar]
- Genetic environment and expression of the extended-spectrum â-lactamase blaPER-1 gene in Gram-negative bacteria. Antimicrob Agents Chemother. 2005;49:1708-13.
- [Google Scholar]
- Transferable production of PER-1 â-lactamase in Pseudomonas aeruginosa. J Antimicrob Chemother. 1995;35:281-94.
- [Google Scholar]
- Prevalence of PER and VEB type extended spectrum beta lactamases among multidrug resistant Acinetobacter baumannii isolates in North-West of Iran. Iran J Basic Med Sci. 2013;16:751-5.
- [Google Scholar]
- Prevalence of PER-1- type extended-spectrum beta-lactamaes in clinical strains of Pseudomonas aeruginosa isolated from Tabriz, Iran. Iran J Basic Med Sci. 2012;15:678-82.
- [Google Scholar]
- Dissemination of class 1, 2 and 3 integrons among different multidrug resistant isolates of Acinetobacter baumannii in Tehran hospitals, Iran. Polish J Microbiol. 2011;60:169-74.
- [Google Scholar]
- Identification of PER-1 extended-spectrum â-lactamase producing Pseudomonas aeruginosa clinical isolates of the international clonal complex CC11 from Hungary and Serbia. FEMS Immunol Med Microbiol. 2008;54:330-8.
- [Google Scholar]
- Clinical and Laboratory Standards Institute (CLSI). Performance standards for antimicrobial susceptibility testing; 21st Informational Supplement. M100-S21. Wayne, PA, USA: CLSI; 2011.
- [Google Scholar]
- Prevalence of Ambler class A and D β-lactamases among clinical isolates of Pseudomonas aeruginosa in Korea. J Antimicrob Chemother. 2005;56:122-7.
- [Google Scholar]
- Identification of epidemic strains of Acinetobacter baumannii by integrase Gene PCR. J Clin Microbiol. 2001;39:8-13.
- [Google Scholar]
- Gene cassette PCR: sequence-independent recovery of entire genes from environmental DNA. Appl Environ Microbiol. 2001;67:5240-6.
- [Google Scholar]
- Molecular cloning: a laboratory manual. (2nd ed). New York: Cold Spring Harbor Laboratory Press; 1989.
- [Google Scholar]
- Identification of plasmids by PCR-based replicon typing. J Microbiol Methods. 2005;63:219-28.
- [Google Scholar]
- Antibiotic susceptibility testing by a standardized single disk method. Am J Clin Pathol. 1966;45:493-6.
- [Google Scholar]
- Molecular epidemiology and mechanisms of carbapenem resistance in Acinetobacter baumannii endemic in New York City. Clin Infect Dis. 2003;37:214-20.
- [Google Scholar]
- Use of an efflux pump inhibitor to determine the prevalence of efflux pump-mediated fluoroquinolone resistance and multidrug resistance in Pseudomonas aeruginosa. Antimicrob Agents Chemother. 2005;49:565-70.
- [Google Scholar]
- Distribution of repetitive DNA sequences in eubacteria and application to fingerprinting of bacterial genomes. Nucleic Acid Res. 1991;19:6823-31.
- [Google Scholar]






